Case Report ID: 24
Title:
Plaquenil-Induced Toxic Maculopathy
Category:
Description:
Plaquenil-induced maculopathy is a form of retinal toxicity that is associated with long-term use of of the anti-inflammatory medication plaquenil.
The drug-induced maculopathy is associated with the following risk factors:
- Cumulative dose level of 1,000 grams of plaquenil
- Patients with existing retinal or macular disease
- Patients over the age of 60
- Patients prescribed with higher doses of medication
- Patients with kidney or liver dysfunction where drug elimination is reduced
- Patients weighing less than 125 pounds on a dosage at or exceeding 400mg/day
Case Report
- A 68-year-old black woman presented for a regularly scheduled surveillance examination to rule out plaquenil-induced maculopathy
- Case history revealed no subjective visual disturbances
- The patient had been examined one year earlier and had been cleared to continue taking plaquenil
Conclusion
Optometrists and ophthalmologists need to be aware of the American Academy of Ophthalmology’s 2011 guidelines on screening for retinal toxicity associated with plaquenil. Newer studies have preferred practice protocols be changed to using spectral-domain OCT technology as the main screening device for detection of early retinal changes in plaquenil toxicity.
History of Present Illness
- Associated Signs and Symptoms: none
Review of Systems
The patient reported that she was in reasonably good health despite a long-term history of lupus. She was taking 200 milligrams of plaquenil per day to treat her lupus and had been on this dose level for the past twenty years. In addition to medication for her lupus, she was also being treated with glyburide for diabetes and an unkown medication for hypertension.
Past, Family and Social History
Non-contributory.
Best-corrected distance visual acuity
- 20/20 in the right eye
- 20/20 in the left eye
Intraocular Pressure Measurements
- 17 mm Hg in the right eye
- 16 mm Hg in the left eye
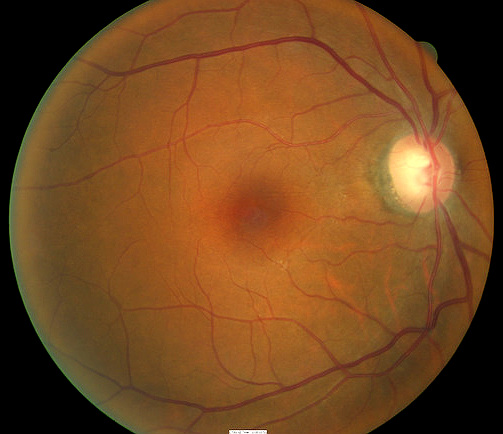
Normal Examination Findings
- Mental status
- General medical observation
- Pupils
- Gross visual fields
- Basic sensorimotor examinaion
- External examination
- Adnexal examination
- External ocular examination with biomicroscopy
- Ophthalmoscopy
Clinical Diagnosis
The clinical diagnosis is a determination based on the knowledge obtained from the patient’s medical history and from the results of the eye examination alone, without the benefit of diagnostic tests or procedures.
The patient’s clinical diagnosis is “high-risk” for developing plaquenil-induced toxic maculopathy based on her medical history and the American Academy of Ophthalmology’s 2011 recommendations regarding the risk factors associated with developing the condition.
(1) The risk of plaquenil-induced toxic maculopathy is associated with a cumulative dose level of 1,000 grams and it usually takes 5-7 years for patients on the standard dose of 250-400 milligrams per day to reach that level.
(2) Based on a dose of 200 milligrams per day for the past twenty years, the patient’s cumulative dose level is 1,460 grams of plaquenil.
- 200 mg x 365 days = 73,000 mg per year
- 73,000 mg x 20 years = 1,460,000 mg cumulative dose level of plaquenil
- 1,460,000 milligrams = 1,460 grams
(3) The cumulative dose level of 1,460 grams is above the 1,000 gram threshold for establishing “high-risk.”
Treatment Plan
To gather the clinical information required to screen for early detection of plaquenil-induced toxic maculopathy, the following diagnostic program could be considered, with emphasis on OCT testing first and visual field analysis second:
- Spectral-domain OCT testing
- Threshold visual field examination with a 10-2 testing strategy
- Fundus autofluorescence (FAF)
- Multifocal electroretinogram (mfERG)
Several macular diseases have clinical signs that mimic the appearance of plaquenil-induced toxic maculopathy.
Differential Diagnoses
Age-related macular degeneration
- Drusen
- Pigment clumping
- Atrophy of retinal pigment epithelium
- Detachment of retnal pigment epithelim
Stargardt’s disease
- Young age
- Family history
- Yellow-white flecks in the macula
Cone dystrophy
- Young age
- Family history
- Photophobia
- Abnormal photopic electroretinogram
Ordering Diagnostic Tests
When the fundus appearance and visual acuity is normal, the following diagnostic tests can help determine the presence of plaquenil-induced toxic maculopathy:
- 10-2 threshold visual field examination
- Electroretinogram
- Retinal laser scan with spectral-domain OCT
- Extended color vision examination
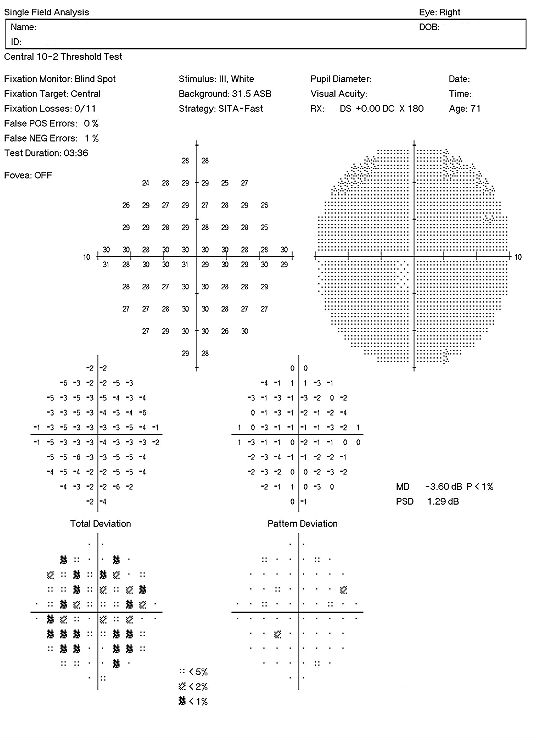
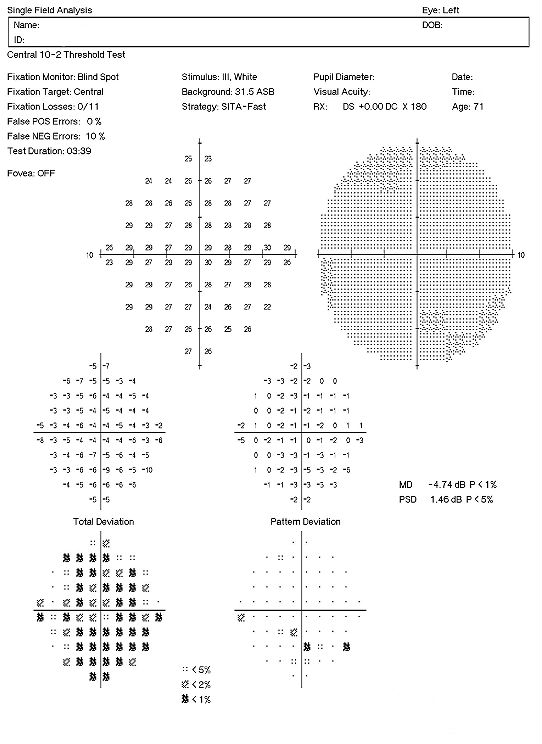
Visual Field Examination
- Automated threshold perimeters meaure the visual field by ploting the threshold luminance value of the patient in various locations in the visual field
- The luminance of the light stimulus is represented by non-specific units of measurement called decibels (dB)
There are several caveats to remember when performing 10-2 visual field examinations on patients taking plaquenil:
- Parafoveal loss of retinal sensitivity may appear before changes are visualized on fundus examination
- Attention should be given to the “Pattern Deviation” to help determine the clinical significance of the test results
- Promptly repeat the examination when central or paracentral visual field defects are observed to determine whether the defects are reproducible
- When visual fields are performed independently, even the most subtle changes or those of uncertain significance should be taken seriously and are an indication for objective testing
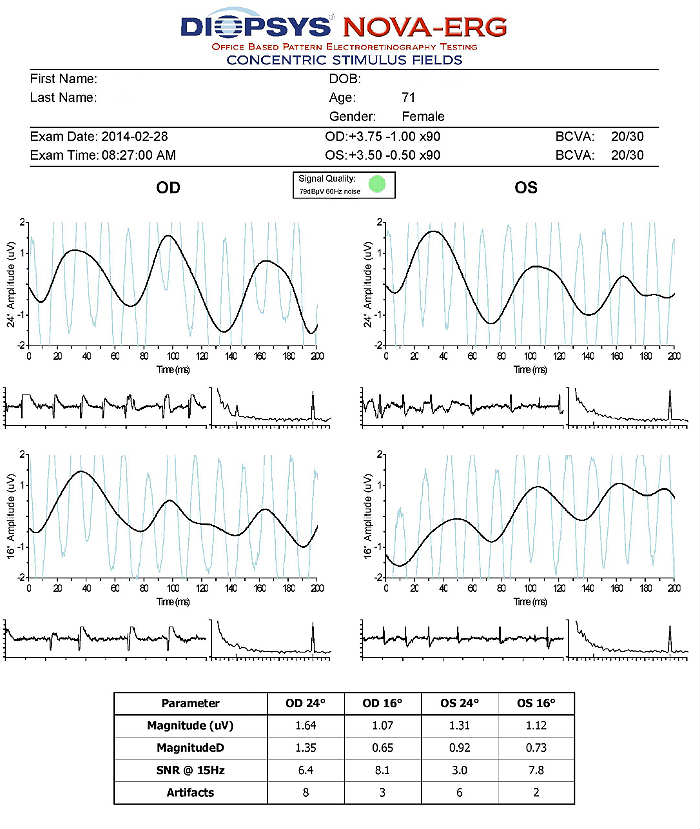
Electroretinography (ERG)
- Electroretinogram testing evaluates the integrity of the retina
- The procedure can be accomplished using the NOVA-ERG Testing Device manufactured by DIOPSYS
- Specialized pattern electroretinogram (PERG) test uses a contrast-reversing pattern for the stimulus to produce information about ganglion cell function
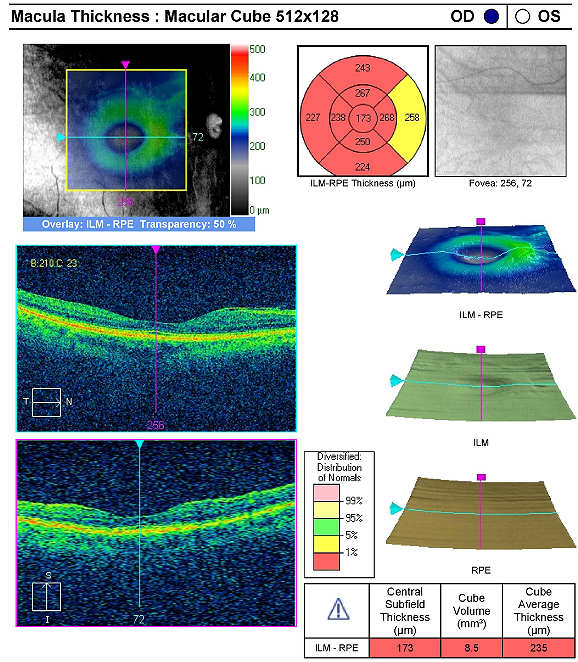
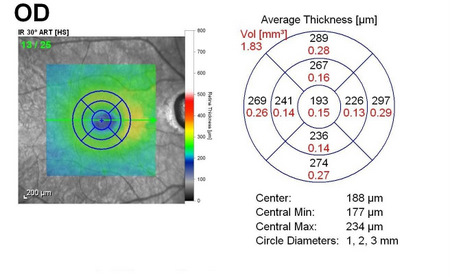
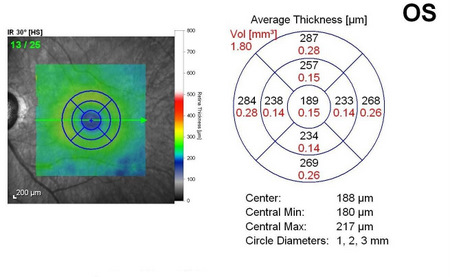
Retinal Scanning Laser
Spectral-domain optical coherence tomography is required to detect early plaquenil-induced structural damage to the macular region. The following instruments can display imaging data in different reports and formats.
- SPECTRALIS OCT by Heidelburg Engineering
- CIRRUS OCT by Carl Zeiss Meditec
- RTVue OCT by Optovue
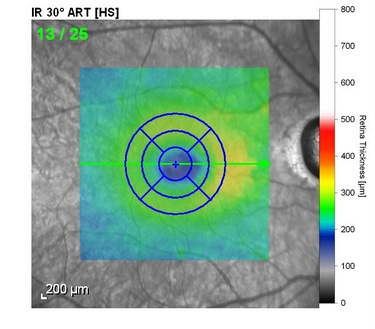
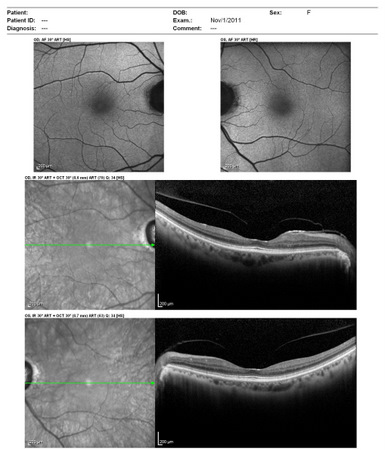
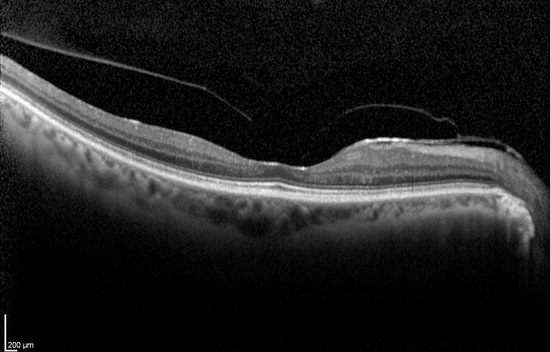
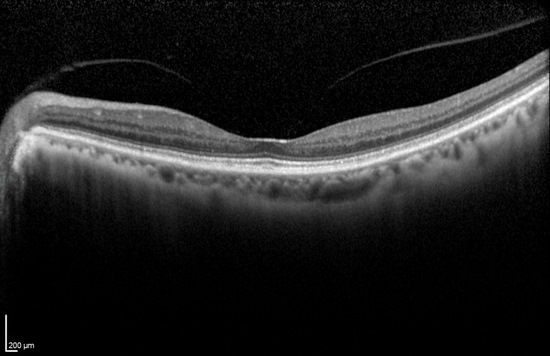
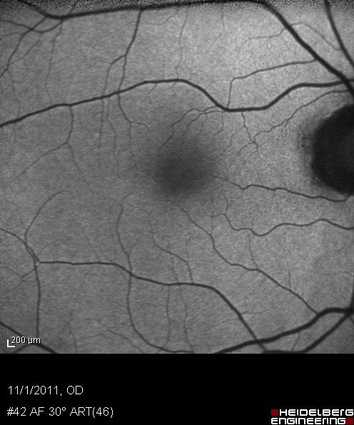
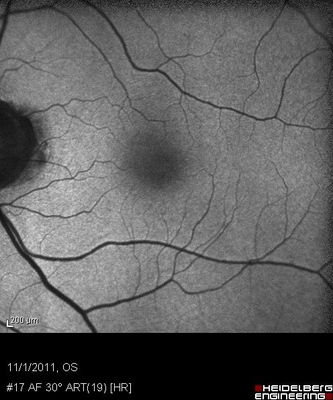
The following scanning laser examinations were obtained on a CIRRUS OCT:
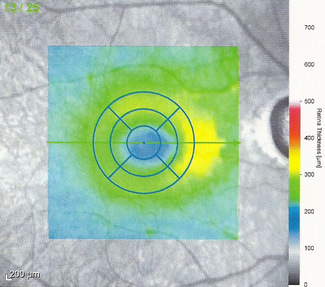
- Parafoveal thinning seen on macular thickness map
- Vitreomacular adhesion revealed on high-resolution scan
Extended Color Vision Examination
An extended color vision examination measures the combined function of the retina, the optic nerve and the intracranial visual pathway and is used clinically to detect or monitor color vision loss due to disease at any of these locations. Extended color vision testing divides people into two groups:
- The first group consists of people with normal color vision and slight color deficiency
- The second group consists of people with moderate or severe color deficiency
One of the most common tests performed in clinical practice is the Farnsworth D15. When performed on Konan Medical’s ColorDx software, the test is presented as fifteen colored bars and one fixed bar on an Android tablet. The hue of each bar has been chosen so that the adjacent bars have approximately equal hue differences. When the bars are arranged in order, they form a hue circle. As a result, errors in hue discrimination can be made across the hue circle.
Patients with normal color vision usually make no error but may make one or two minor transposition errors, as do those with mild color vision deficiency. Patients with more advanced color deficiency make some or more of the following errors:
- They place colors that lie on the opposite side of the hue circle next to colors that lie on their confusion locus (e.g., diametrical errors)
- Two or more diametrical crossing errors indicates a “failed” test
- If diametrical crossing errors are made, the orientation of the crossing produces a diagnosis of protan, deutan, or tritan color vision deficiency
Plaquenil-induced toxic maculopathy was first described in 1967. Because the retinal damage from plaquenil is usually irreversible if present for a more prolonged period of time, it is important to detect the disease in its early stages.
All of the diagnostic test results confirmed the initial diagnosis of early retinal toxicity from plaquenil therapy. Photographic documentation of the macula would be an additional medically necessary test that could be obtained at any follow up evaluation.
According to Current Procedural Terminology, when eye doctors perform ophthalmological examinations, the complexity of medical decision-making is not separated from the examining techniques used. As a guideline to assist eye doctors in enhancing their medical decision-making skills, consider that the complexity of medical decision-making involves three components.
The first component concerns the number of possible diagnoses and treatment options that must be considered. Determination of the diagnosis of plaquenil toxicity involves the determination of structural damage to the retina, typically prior to any functional loss of vision.
The second component concerns the amount and complexity of medical records and diagnostic tests that have to be obtained, reviewed and analyzed. In addition to an eye examination, this visit required the review and analysis of a subjective refraction, a retinal laser scan, and a threshold visual field examination.
Third, the complexity of medical decision-making is affected by the risk of significant complications and/or morbidity associated with plaquenil toxicity and the risks involved in any treatment options. This patient’s retinal condition was classified as a significant problem where the risk of significant central vision loss without treatment was possible. In addition, the treatment plan will involve coordination of care with the patient’s primary care physician as well as continued monitoring of her retinal condition.
Treatment Guidelines
Plaquenil induced retinal toxicity is reversible if detected early and plaquenil dosage altered or the medication discontinued. Even suspicious early plaquenil toxicity warrants communication with the patient’s treating physician, and meaningful coordination of care would include a report even if the findings are normal. In most all cases of toxicity, the medication will be discontinued.
There is no local (retinal) treatment for early plaquenil toxicity so no referral to a retinal specialist is warranted.
Treatment Program
The patient was referred back to her treating physician with a complete report of the ocular findings and recommendations to consider alteration of her current therapy.
Next visit scheduled for 3 months.
Discussion
This case demonstrates several key aspects of monitoring ocular side effects of systemic medication. First and foremost, it demonstrates the key role of eye doctors in the detection of serious, sight threatening complications of systemic medications. Second, it shows how modern technology has changed the ability to detect early and subtle pathology associated with these medications. For years, eye doctors have relied on threshold visual field testing to detect functional vision changes with plaquenil use, rarely finding any signs of toxicity except in advanced cases where vision symptoms are already manifest. Spectral domain OCT testing provides eye doctors a highly sensitive and specific tool to detect retinal changes long before subjective vision loss occurs.
Based on patient history, the nature of the presenting problem and my own clinical judgment, this patient needed an evaluation of the complete visual system.
- Perform the eye examination that is medically necessary
- Provide the diagnostic tests or services that are medically necessary
- Properly document the services provided
- Code from the documentation
- Report the services to the payor
Physicians Quality Reporting System
PQRS Measure 130: Current Medications with Name, Dosage, Frequency and Route Documented – CPT code G8427
This measure applies to patients 18 years and older. The measure should be reported on the day of the examination and can be used with any diagnosis code.
PQRS Measure 226: Patient screened for tobacco use and identified as a non-user of tobacco – CPT code 1036F
This measure applies to patients 18 years and older. The measure should be reported on the day of the examinaton and can be used with any diagnosis code.
Multiple Procedure Payment Reduction
Effective January 1, 2013, there is a small reduction in payment from Medicare if certain multiple procedures are billed on the same day. The fee for the technical component of the diagnostic test for the second and subsequent tests will be reduced by 20%. The second diagnostic test and subsequent tests should be reported with a -51 modifier. Professional services such as gonioscopy, extended ophthalmoscopy, serial tonometry and provocative glaucoma testing are excluded from this policy. Visual evoked potential testing is excluded from this policy.
Modifier 51
This modifier is used to identify the secondary procedure or when multiple procedures are performed on the same day by the same provider. List the major primary procedure first and append the modifier to the subsequent procedures. The primary procedure is the one with the highest dollar value.
| Diagnosis Code | Procedure Code | Modifier | Quantity | Payor | Amount Allowed |
| H35.383 - Toxic maculopathy, bilateral | 92014 - Medical eye examination | 1 | Medicare | 127.03 | |
| Z79.899 - Other long term (current) drug therapy | 92275 - Electroretinography | 1 | Medicare | 167.58 | |
| H35.383 - Toxic maculopathy, bilateral | 92083 - Visual field examination | 51 | 1 | Medicare | 58.45 |
| H35.383 - Toxic maculopathy, bilateral | 92134 - Retinal laser scan | 51 | 1 | Medicare | 42.19 |
| H35.383 - Toxic maculopathy, bilateral | 1036F - Patient screened for tobacco use | Medicare | 0.00 | ||
| H35.383 - Toxic maculopathy, bilateral | G8427 - Current medications documented | Medicare | 0.00 | ||
| Total | $228.00 |
H35.381
Toxic maculopathy, right eye
H35.382
Toxic maculopathy, left eye
H35.383
Toxic maculopathy, bilateral
362.55
Toxic maculopathy
92015
Refraction
92250
Fundus photography
92083
Visual field examination
92283
Color vision examination
92134
Retinal laser scan
92225
Extended ophthalmoscopy
92275
Electroretinography




 Print | Share
Print | Share