ICD-10 Diagnosis Code:
T86.890 — Other transplanted tissue rejection
Title
Amniotic Membrane Allograft Rejection
Category
Complications Of Transplanted Organs And Tissues
Description
Immunologic rejection of the amniotic membrane is one of the most common causes of allograft placement failure.
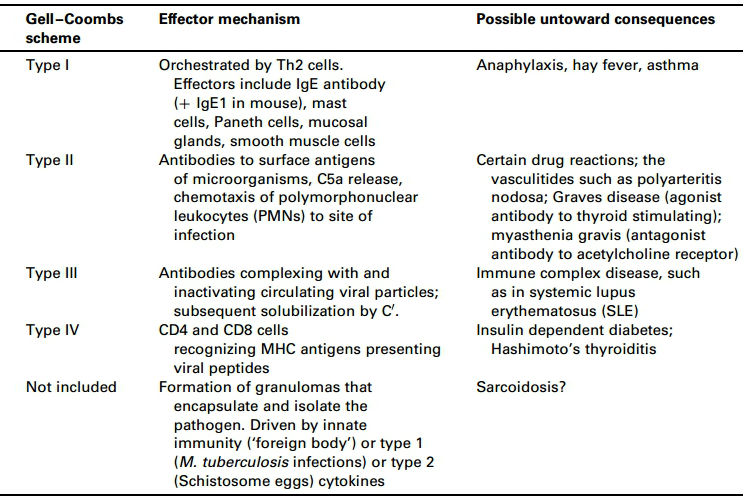
Most amniotic membrane allograft rejections are a type 2 allergic reaction that involves the interaction of immunoglobulins with foreign or autoantigens closely associated with cell membranes. In this condition the antigen is the amniotic membrane allograft.
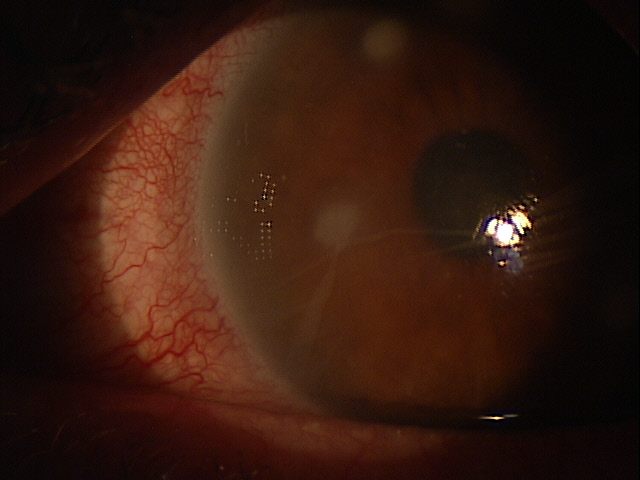
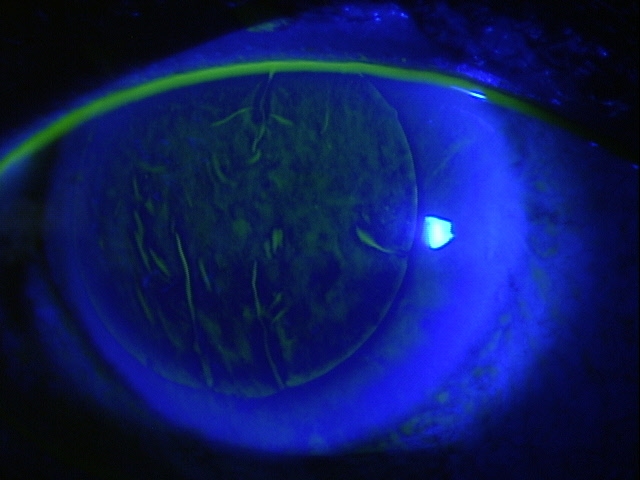
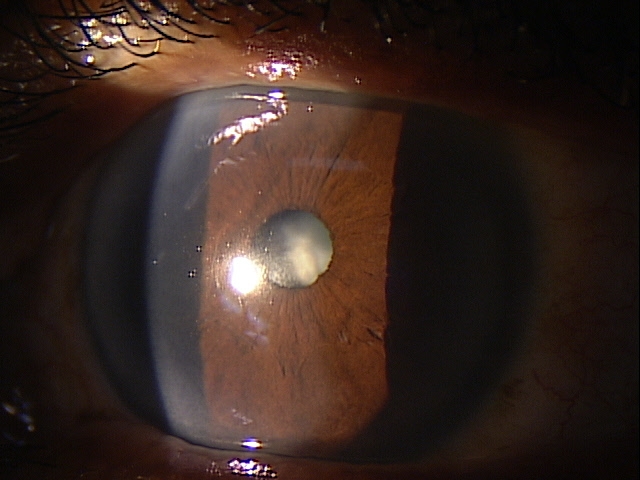
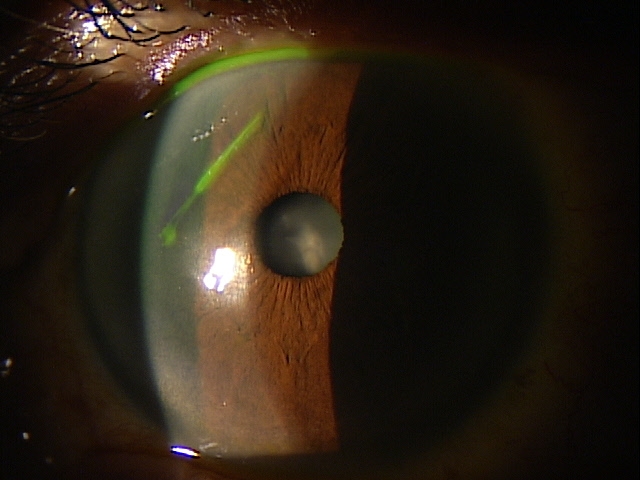
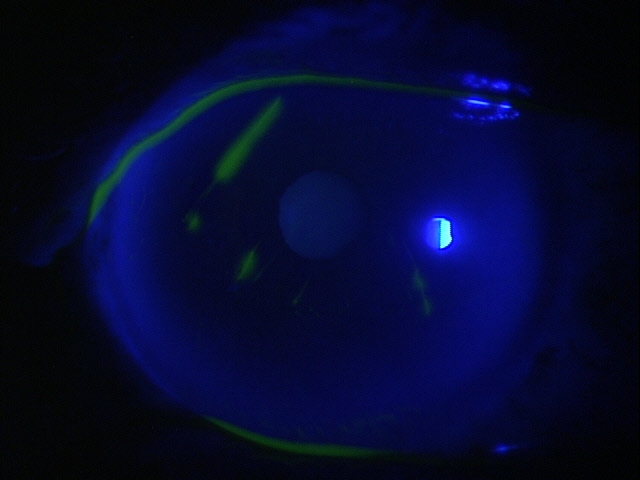
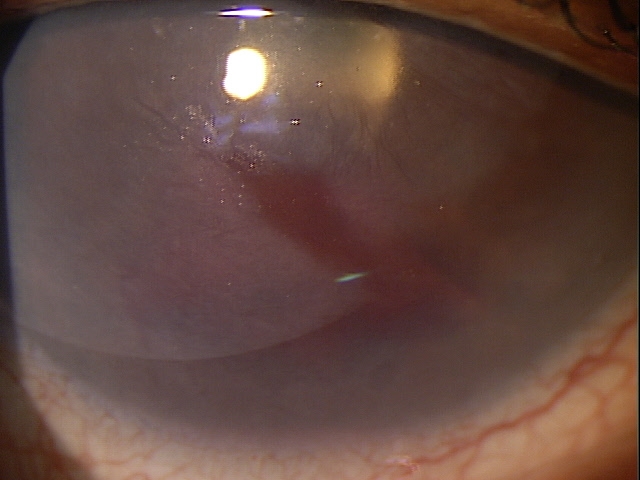
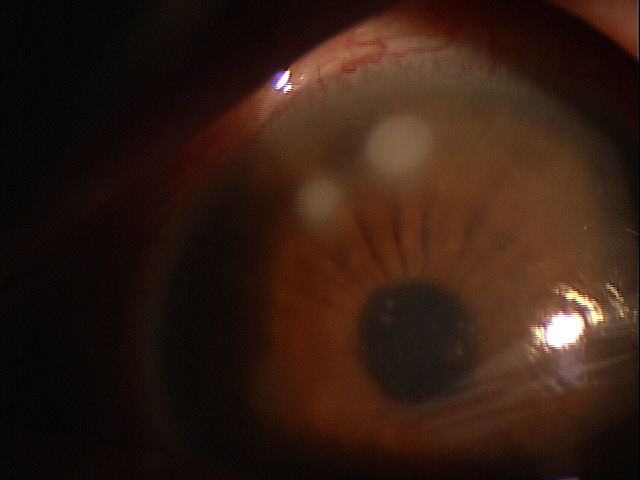
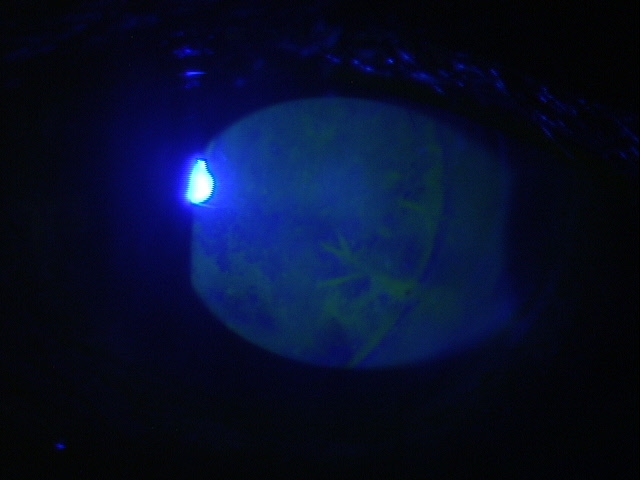
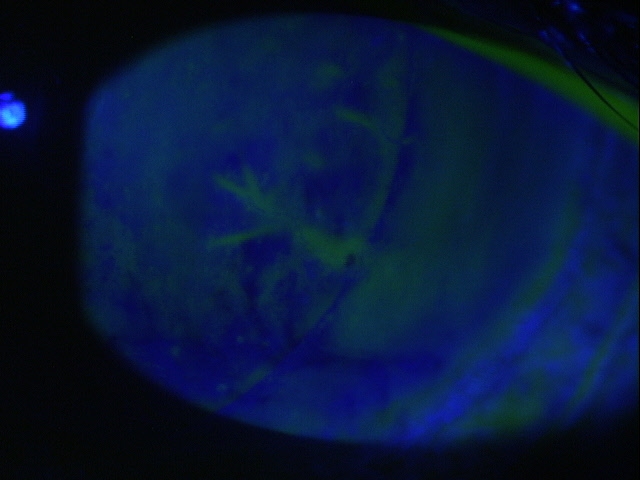
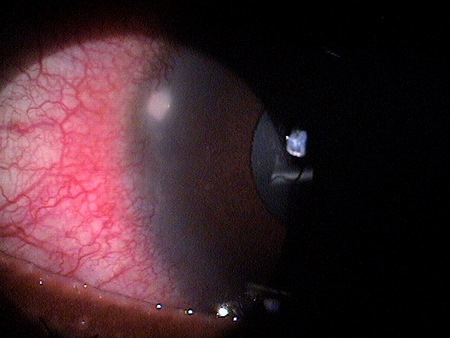
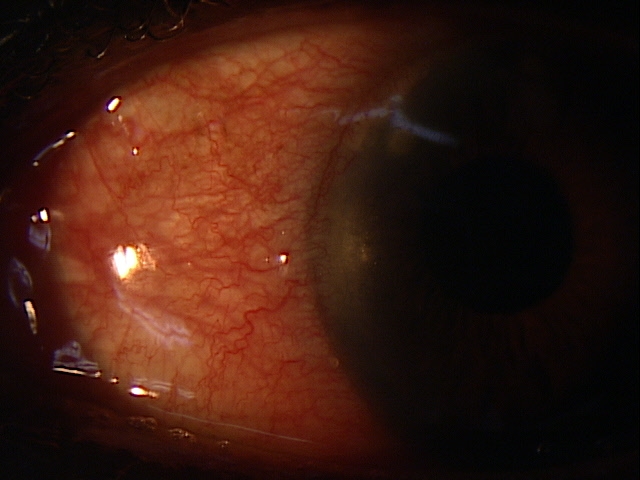
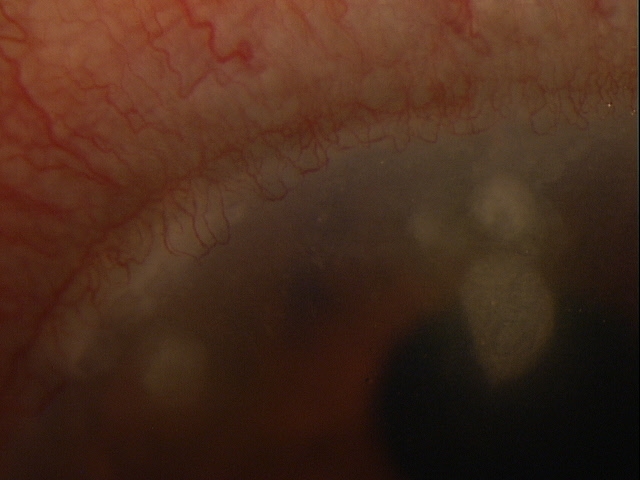
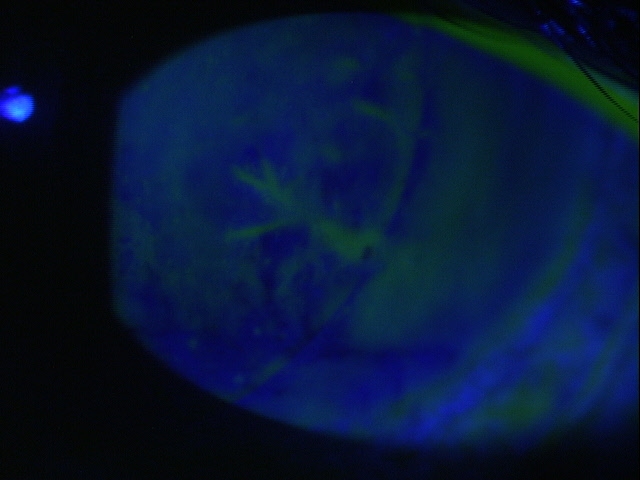
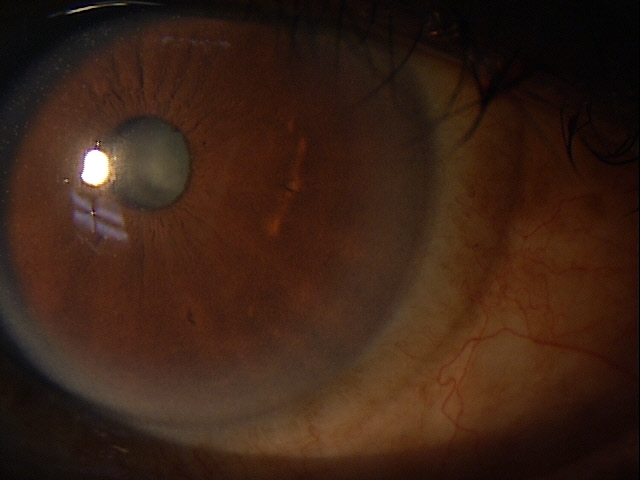
In rejection episodes involving dehydrated membranes, there is usually a coarse punctate epitheliopathy that is present when the cornea is examined with fluorescein. If there are folds in the membrane, there may be linear-shaped epithelial rejection lines on the cornea that correspond to the folds. Early symptoms include a foreign body sensation and mild ocular discomfort. Severe reactions can include mid-peripheral corneal infiltrates, conjunctival hyperemia, eyelid edema, increased intraocular pressure, and significant eye pain.
When using cryopreserved membranes, the rejection epitheliopathy is more diffuse, but the anterior chamber and adnexal reactions are similar. Because some patients notice discomfort associated with the symplepharon ring, it can be difficult to differentiate discomfort from the ring vs. discomfort from an immunologic response.
Amniotic membrane allograft rejection refers to a specific immunologic response of the host to the donor placental tissue. Although the goal of amniotic membrane tissue preparation is to produce a product that is antigen neutral, all currently available amniotic membrane allografts are capable of producing an immune reaction in individual patients because they contain “non-self” low molecular weight peptides or residual DNA from the donor.
Structural Damage to the Eye
- Allograft is recognized as foreign and IgG or IgM antibodies are formed which react with and activate the serum complement enzymatic cascade
- Corneal stromal cell lysis may result from complement activation and from recruitment of leukocytes
 |
 |
Functional Damage to the Eye
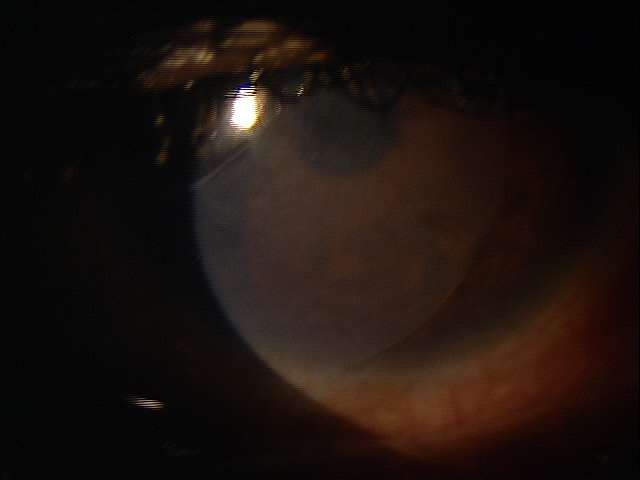
- Decreased vision secondary to corneal, anterior chamber or retinal complications
 |
 |
The main goal in the diagnostic evaluation of a patient with amniotic allograft membrane rejection is to accomplish the following:
- Distinguish the response from other non-immune allograft failures
- Relieve ocular pain and discomfort
- Prescribe a treatment program
To obtain the information required to determine a clinical diagnosis of tissue rejection and to prescribe a treatment plan, the following service components of a medical eye examination should be performed.
- Patient history
- General medical observation
- Adnexal examination
- External ocular examination with biomicroscopy
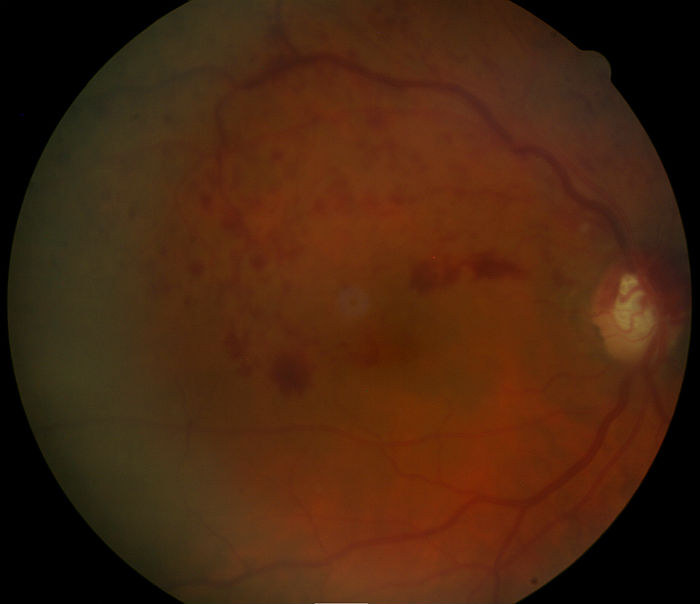
- Ophthalmoscopy
Patient History
Patients with amniotic membrane allograft rejection usually present with abnormal signs and symptoms within hours of the placement procedure. Some or all of the following symptoms may be present during a rejection episode:
- None
- Localized eye pain
- Orbital pain
- Foreign body sensation
- Hyperlacrimation
- Burning
- Redness
- Itching
- Photophobia
- Eyelid edema
- Corneal edema
- Stromal infiltrates
- Epithelial rejection line
- Coarse punctate epitheliopathy
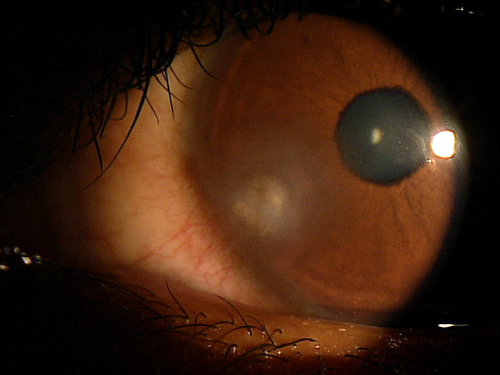
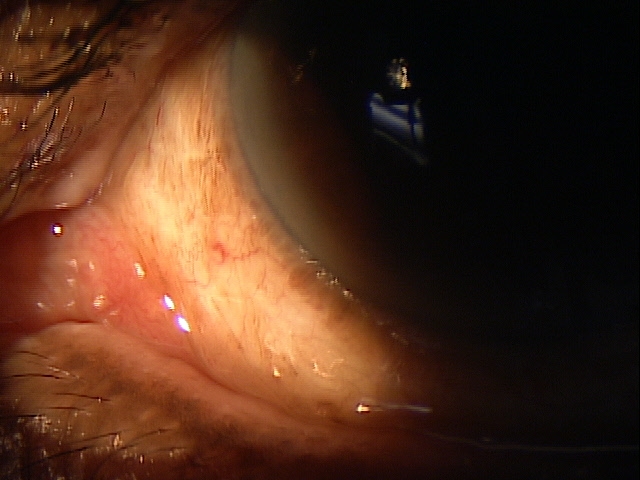
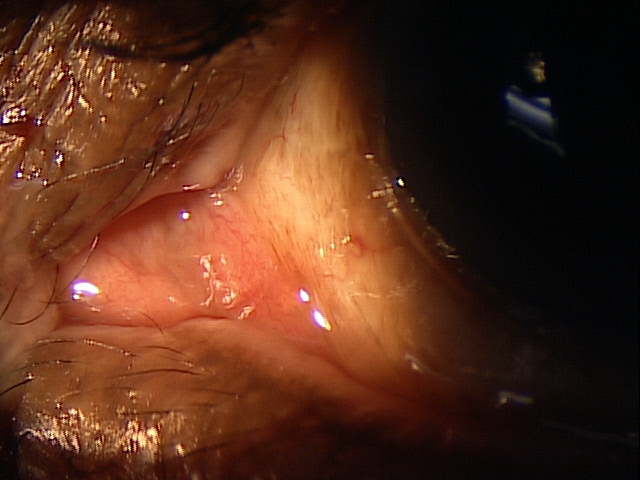
Adnexal Examination — PATIENT 1
- Eyelid edema
- Eyelid erythema
- Eyelid tenderness
 |
 |
 |
 |
Clinical Appearance of the Eyelid
|
|
Clinical Appearance of the Eyelid
|
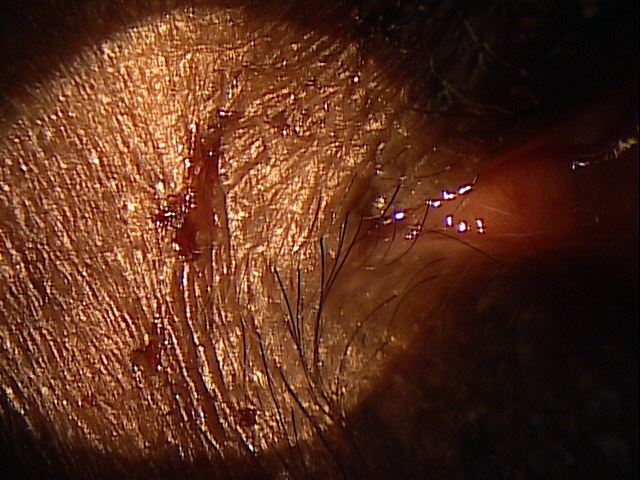 |
|
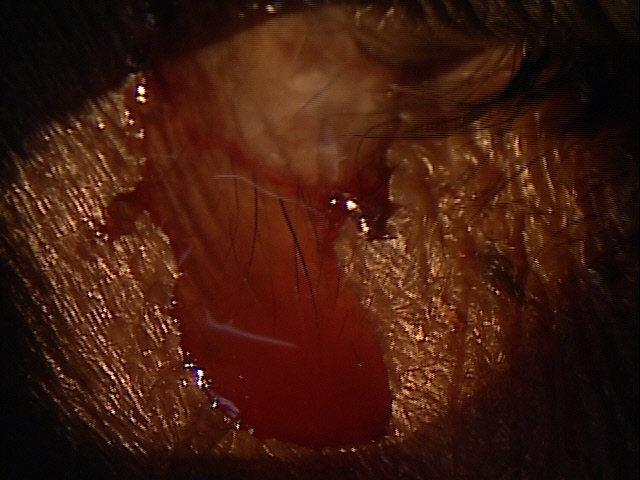 |
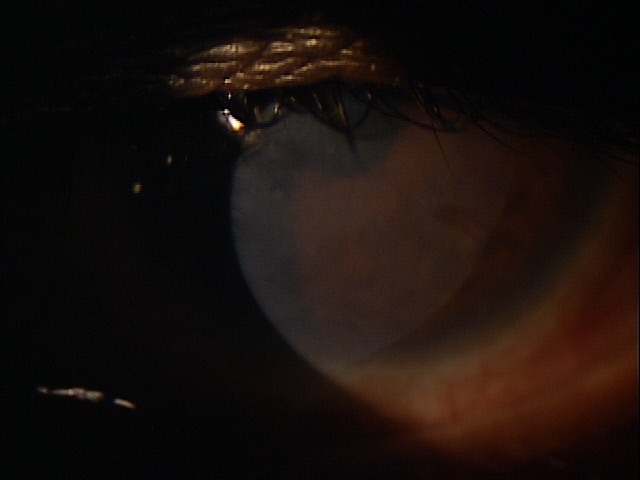
Clinical Appearance of the Tear Film
|
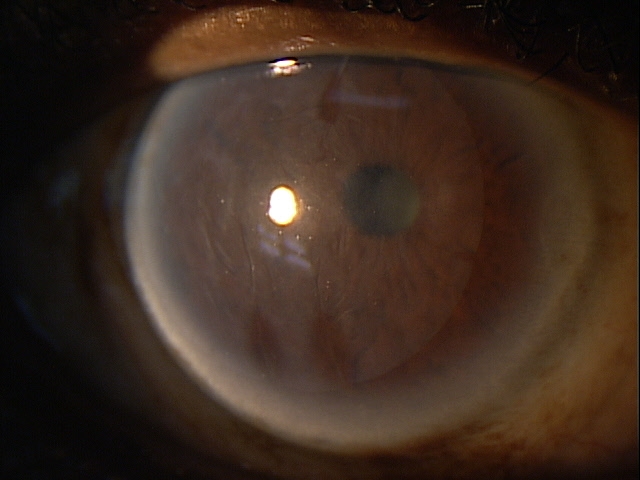
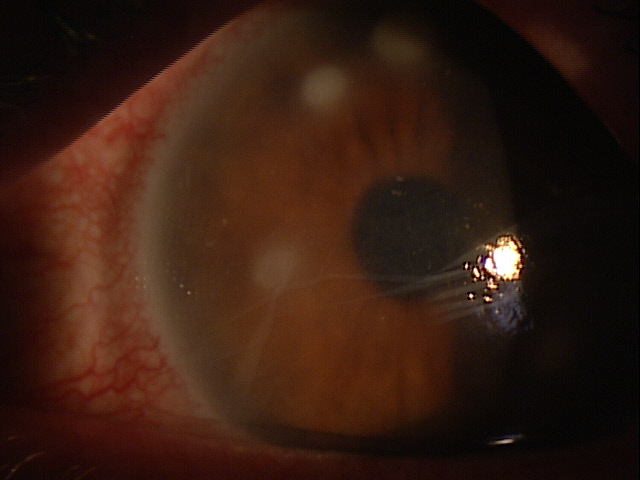
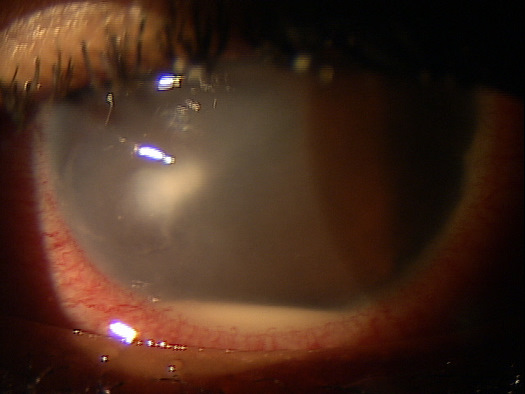
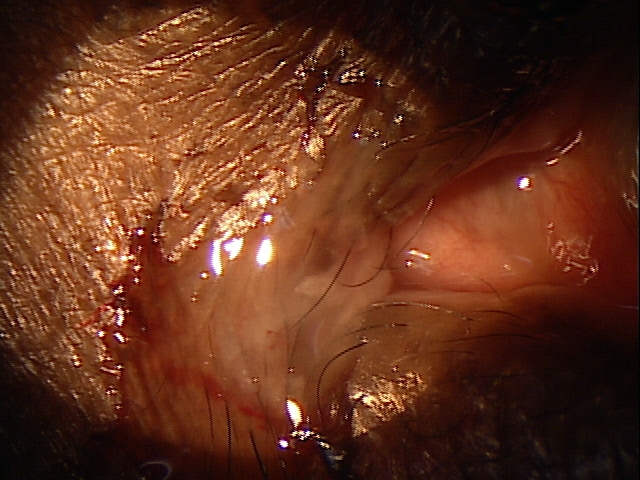
External Ocular Examination with Biomicroscopy — PATIENT 2
- 70-year-old Black woman with open-angle glaucoma
- Preoperative diagnosis: Keratoconjunctivitis
- Postoperative symptoms: Severe foreign body sensation
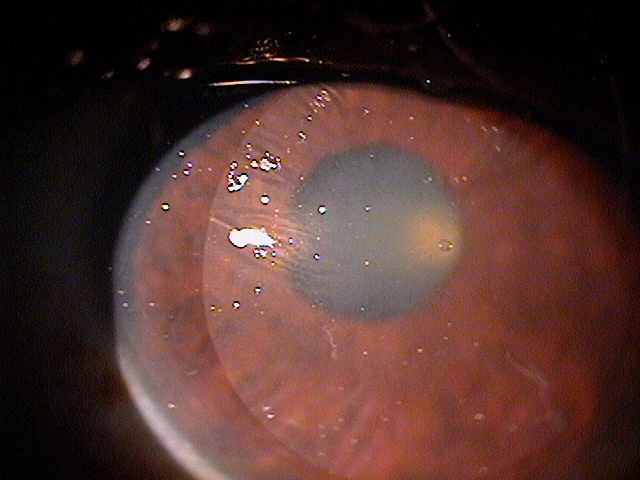
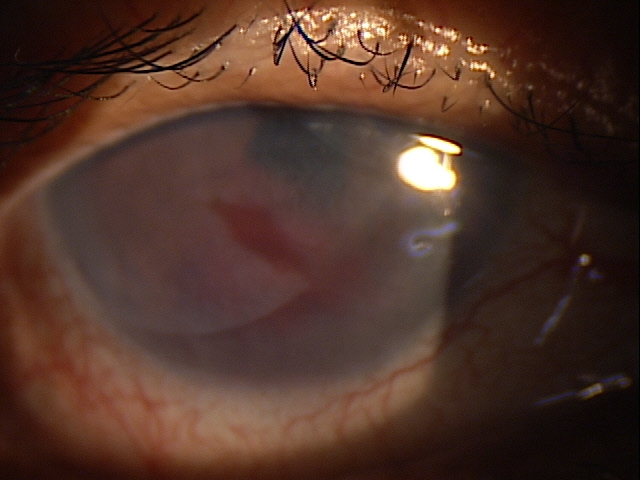
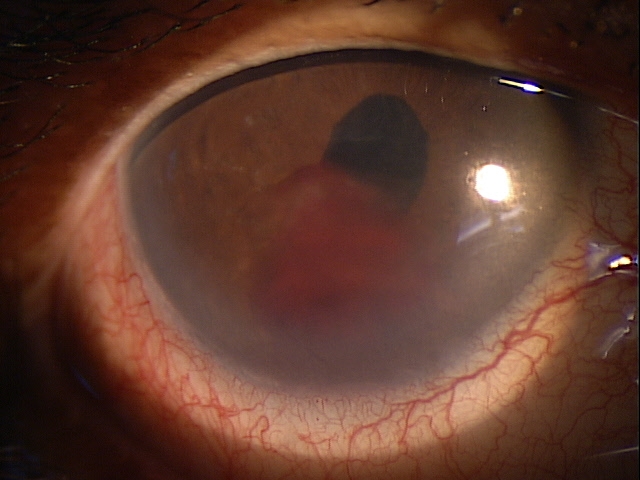
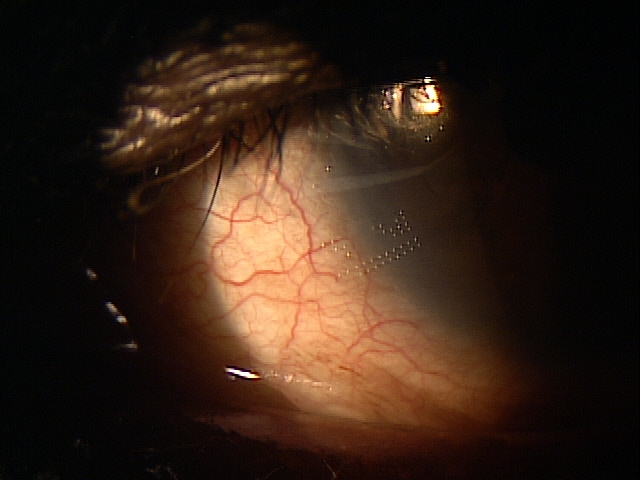
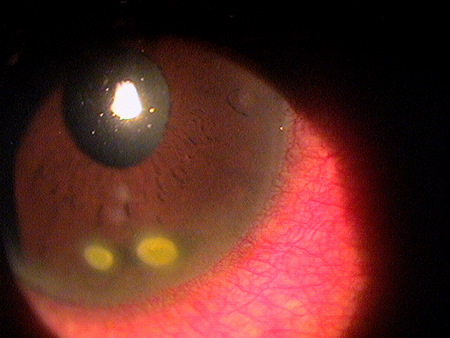
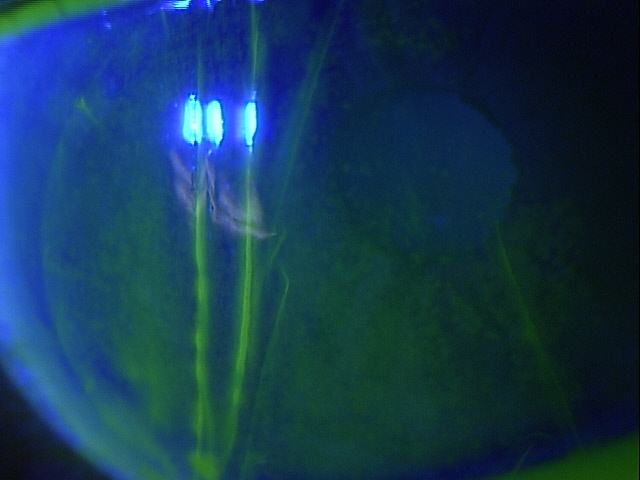
External Ocular Examination with Biomicroscopy — PATIENT 3
- xxx
- xxx
- xxx
- xxx
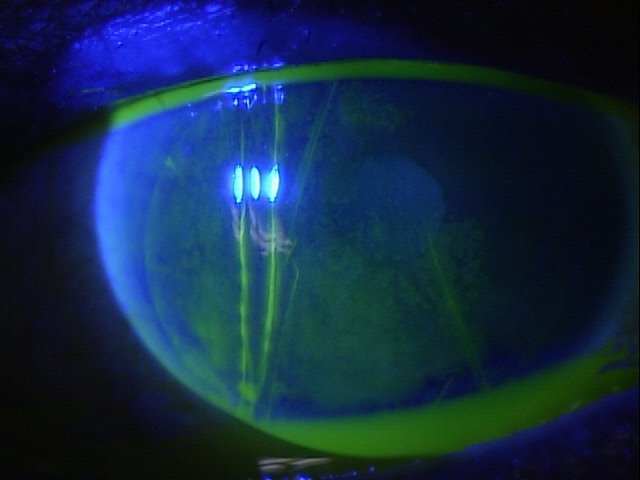 |
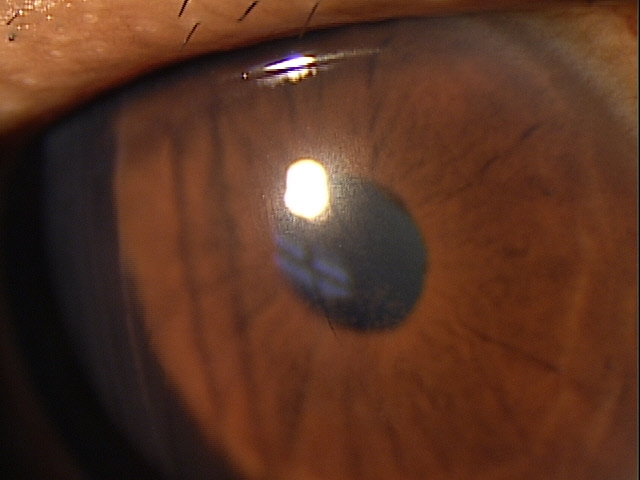 |
 |
The goals of treating amniotic membrane allograft rejections include the following:
- Improve patient symptoms
- Reduce chronic gland inflammation
- Prevent damage to the ocular surface
- Prevent meibomian gland dropout
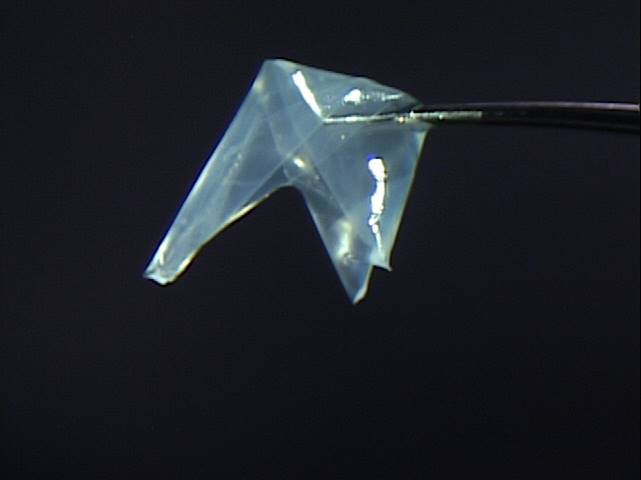
Mechanical Treatment
- Remove the membrane
- Bandage contact lenses with prophylactic antibiotic eye drops can be used in moderate to severe presentations
Pharmocologic Treatment
- Non-preserved artificial tears can be used to lubricate the cornea and relieve ocular discomfort
- Topical steroid eye drops can modify or reduce the number of mucus receptor sites by inhibiting inflammatory cells and fibroblasts at the base of the corneal filament
- Topical 5% hypertonic solution reduces the formation of corneal filaments by inhibiting receptor site formation and increasing the tear-to-mucus ratio of the precorneal tear film
- Acetylcystene 10% can help eliminate the filament formation in severe presentations
Aggressive eradication of infection is the most important step in treating an amniotic membrane rejection, which may involve around-the-clock use of antibiotic eye drops. Cycloplegic (dilating) drops can be used for relief from light sensitivity.
Topical corticosteroids should be used every hour while awake and as frequently as possible at night for 2-3 days, followed by every 2 hours while awake. Steroid ointment may be used at bedtime. Therapy should be continued until signs of rejection resolve.
In all cases of rejection, intraocular pressure should be monitored closely, especially when frequent corticosteroids are used. If necessary, elevated intraocular pressure should be controlled by topical medications to prevent glaucomatous reactions.
1. Reinhard T, Larkin F. Essentials in Ophthalmology: Cornea and External Eye Disease. Springer 2010 ed.
2. He H, Zhang S, Tseng S. Amniotic Membrane’s Therapeutic Action. Rev Optom, 8 Oct 2014. https://www.reviewofophthalmology.com/article/amniotic-membranes-therapeutic-action. Last accessed July 31, 2016.
2. Mastroda, K. Amniotic Membrane in Ocular Surface Disease. Optometry Times 20 Dec 2013. http://optometrytimes.modernmedicine.com/optometrytimes/content/tags/amniotic-membrane/amniotic-membrane-ocular-surface-disease. Last accessed August 1, 2016.
3. Bronner, A. Learn To Avoid Rejection. Rev Optom. 16 June 2010. https://www.reviewofoptometry.com/article/learn-to-avoid-rejection. Last accessed August 1, 2016.
4.0
996.89
Complications of other specified transplanted organ
92285
External ocular photography
92286
Specular endothelial microscopy
92025
Corneal topography
92132
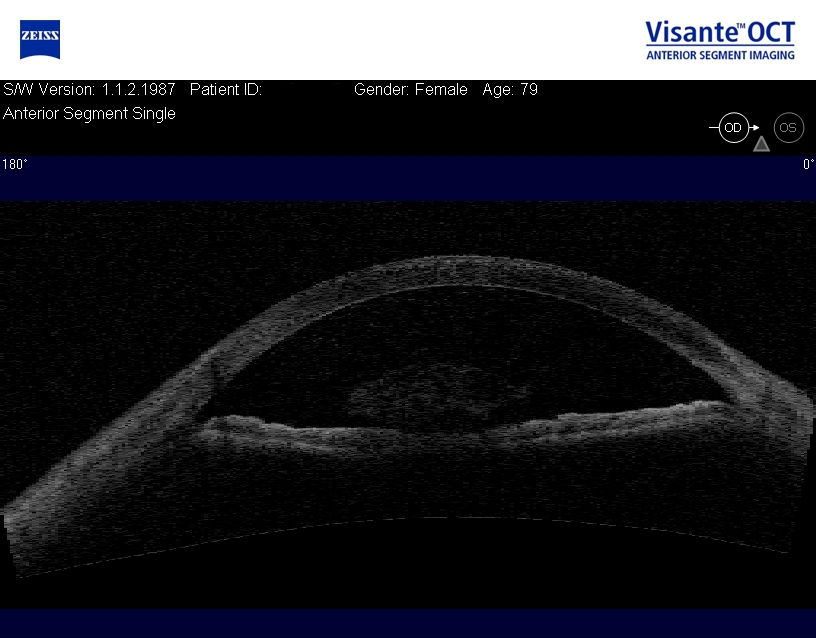
Anterior segment imaging




 Print | Share
Print | Share